Dr. SANJANA RAO
Dr. SANJANA RAO, Dr. DEVENDRA MAHESHWARI
Semi Finals
Abstract
To report the outcomes of a new ab interno technique of sulcus insertion of the AADI tube. Non-comparative, interventional case series. Nine eyes of nine patients who underwent sulcus implantation by the new technique between 2019 and 2020 with a follow-up of at least 3 months were included. Control of intraocular pressure (IOP), number of pressure-lowering medications, visual acuity and surgical complications were recorded. The mean age was 57.22 ± 45–66 years The IOP was reduced from a preoperative mean of 28.33 ± 9.80 to 11.56 ± 2.65 mm Hg (p = 0.001) at 3 months. The mean number of preoperative medications for IOP control was reduced from 3.0 ±0.7 to 0.4 ± 0.9 (p <0.05).There were no intraoperative complications noted. This technique of placement of the silicone tube in the posterior chamber sulcus is a safe and effective alternative to existing procedures of sulcus placement limiting multiple blind entries and thereby, preventing damage to adjacent angle structures
Full Text
Abstract :
Aim : To report the initial clinical outcomes of a new technique of insertion of the silicone tube of the AADI in the posterior chamber sulcus
Methods: Non-comparative, interventional case series. Nine eyes of nine patients, with a follow-up of at least 3 months who underwent sulcus implantation of the AADI tube by the new technique between 2019 and 2020 were included. Control of intraocular pressure (IOP), number of pressure-lowering medications, visual acuity and surgical complications were recorded.
Results : Nine eyes of nine patients (mean (range) age 57.22 (45–66) years) were included in the study. The IOP was reduced from a preoperative mean (SD) of 28.33 (9.80) to 11.56 (2.65) mm Hg at 3 months. The mean (SD) number of preoperative medications for IOP control was reduced from 3.0 (0.7) to 0.4 (0.9) medications in the same period. There were no intraoperative complications noted.
Conclusion: This technique of placement of the silicone tube in the posterior chamber sulcus is a safe and effective alternative to existing procedures of sulcus placement limiting multiple blind entries and thereby, preventing damage to adjacent angle structures
Introduction
Glaucoma drainage devices (GDD) have attained popularity as the procedure of choice in refractory glaucomas.1 The commonly used GDDs today include the Ahmed glaucoma valve (AGV; New World Medical, Rancho Cucamonga, CA, USA), Baerveldt glaucoma implant (BGI; Advanced Medical Optics, Santa Ana, CA) and recently, the Aurolab aqueous drainage device (AADI; Aurolab, Madurai, India).The AADI implant is a newer non-valved GDD derived from the Baerveldt prototype which has demonstrated good long term control of IOP in numerous recent studies.2-7
The commonly reported complications of any GDD include postoperative hypotony, excessive capsular fibrosis, tube erosion and plate migration, strabismus and rarely endophthalmitis.8 However, the most significant long term complication of tube shunt insertion into the anterior chamber is corneal endothelial damage. Accurate placement of the tube away from the cornea is hence essential. The techniques of tube insertion into the ciliary sulcus and pars plana have been increasingly performed in pseudophakic and aphakic eyes respectively to evade this complication. Although pars-plana vitrectomy is relatively safe when performed by experienced hands, vitrectomy and tube insertion into the posterior segment carries significant risks of retinal detachment, endophthalmitis, vitreous haemorrhage and vitreous incarceration in the tube lumen.10,11
The ciliary sulcus is an anatomic space of potential importance for tube insertion especially in pseudophakic patients due to its expansion in anterior–posterior depth following cataract extraction owing to the smaller diameter and thickness of IOL of the intraocular implant as compared to the crystalline lens.9 Studies have reported tube placement in the ciliary sulcus to be a safe and effective procedure offering adequate IOP reduction and reduced endothelial cell loss.9,12 The commonly employed technique described for sulcus placement of the GDD tube involves a relatively blind procedure of entry 1.5 to 2.5 mm away from the limbus directed towards the sulcus with prior insertion of viscoelastic.9,12 However, bleeding in the anterior chamber, trauma to the iris, ciliary body and zonules, intraocular lens (IOL) displacement and inaccurate entry behind the IOL are potential complications to this procedure hence, needing longer learning curve.
In this article, we report the initial clinical outcomes of a novel technique of sulcus placement of the silicone tube of the Aurolab aqueous drainage implant (AADI) in pseudophakic patients as an alternative to the conventional method.
Patients and methods
The retrospective case series was conducted in accordance with the tenets of the Declaration of Helsinki of 1975 after getting approval from the Institutional Review Board and ethical clearance from the Ethics Committee. The nature, risks and possible adverse consequences of the procedure were explained to patients and consent taken from them. A single surgeon (DM) performed the procedure at the centre (Aravind Eye Care System, Tirunelveli, Tamil Nadu, India).
The charts of patients more than 16 years of age undergoing AADI insertion in the posterior chamber sulcus by the new technique and having at least 3 months of follow-up between July 2019 to January 2020 were reviewed. All patients with pseudophakia or those who underwent cataract extraction and intraocular lens placement at the time of the AADI implantation were included. Patients were selected for AADI surgery on the basis of uncontrolled intraocular pressure (IOP) after maximally tolerated medical treatment. Primary AADI implantation with or without combining phacoemulsification with intraocular lens implantation was carried out in all patients. The reason for the same was owing to the scarred conjunctiva from previous small incision cataract surgery with PCIOL implantation, secondary glaucomas such as neovascular glaucoma (NVG), Iridocorneal endothelial syndrome (ICE) syndrome and extensive conjunctival scarring after retinal detachment surgery.
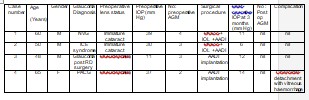
The following data were collected for each eye pre-operatively name, age , gender, glaucoma diagnosis and lens status. The visual acuity, IOP, number of pressure-lowering medications were recorded both pre and post operatively. The intraoperative and post- operative complications were also noted. Successful IOP control was defined by Goldmann tonometry readings between 5 and 20 mm Hg.
Surgical technique
A 350 mm2 Aurolab Aqueous Drainage Implant (AADI; Aurolab, Madurai, India) was used for the procedure. The surgery was performed under peribulbar anaesthesia using a mixture of 1% Lignocaine and 0.5% Bupivacaine. A fornix-based conjunctival peritomy was created spanning 4-5 clock hours in the supratemporal quadrant. The Tenon’s capsule was dissected by blunt dissection to expose the underlying sclera and dissection was carried out till the equator. The superior and lateral recti muscles were sequentially isolated, and the lateral expansions of the AADI device was placed beneath adjacent muscle bellies. The plate of the implant was then anchored to the sclera 9 to 10 mm posterior to the limbus using two interrupted sutures of 8-0 nylon (monofilament polyamide black, Ethilon; Ethicon, Johnson & Johnson, India ) through the fixation holes. After priming the implant using balanced salt solution, the tube of AADI was occluded tightly near tube plate junction using two 6–0 vicryl sutures (Braided-coated polyglactin 910 violet, Ethicon, Johnson & Johnson, India).
The absence of flow through the tube was confirmed by irrigation of balanced salt solution into the tube via a 27-gauge cannula. The tube was then trimmed in a bevel-up fashion to have an intraocular segment of approximately 4 mm . A rectangular scleral flap with dimensions 4 x 4 mm was made and raised. Based on the intended site of implantation, using an ab interno approach, a 21 G vein needle (Infusion set -Type 500, JMS Singapore Pte Ltd) was used to make an entry into the anterior chamber thorough the opposite limbus. The needle was directed to the sulcus by crossing the pupil entering the plane under the iris thereby, emerging out through the bed of the scleral flap 2 to 2.5 mm from limbus tunnel partially. The anterior chamber was well formed using highly cohesive viscoelastic throughout the procedure. Through the exposed hollow end of the bevel of the needle (diameter – 800 microns ), the tube (external diameter – 640 microns) was then inserted so as to be accommodated into the hollow bevel and was guided into the sulcus by withdrawing the needle along the direction of its insertion. The scleral flap was sutured with four 8-0 vicryl sutures (Braided-coated polyglactin 910 violet, Ethicon, Johnson & Johnson, India). The conjunctiva was then closed with the same 8-0 vicryl suture (Braided-coated polyglactin 910 violet, Ethicon, Johnson & Johnson, India) in a continuous fashion. Phacoemulsification was carried out as indicated after securing the plate via a temporal clear corneal section. A foldable PCIOL was implanted in the bag and the section was sutured with 10-0 nylon (monofilament polyamide black, Ethilon; Ethicon, Johnson & Johnson, India).
Results
Nine eyes of nine patients of mean age 57.22 ± 7.85 years; (range, 45–66 ) who underwent the new surgical procedure within the study period meeting the entry criteria were included. The mean follow-up period was 3.90 ± 0.97 months (range 3.07– 6.17) and the male: female ratio was 7:2.
The pre-op and post op parameters are as described in Table 1 . Six of nine patients (66 %) had secondary glaucoma namely neovascular glaucoma (50%),ICE syndrome, glaucoma post retinal detachment surgery and pseudoexfoliation glaucoma in 1 patient each (16%). Primary glaucomas (1 POAG and 2 PACG eyes) were seen in 3 of 9 patients (33%) respectively. Five of nine (55%) patients were pseudophakic and hence underwent primary AADI implantation. Four of nine patients (44%) had cataract and underwent phacoemulsification with intraocular lens implantation combined with primary AADI implantation.
The IOP was reduced from a preoperative mean of 28.2 ± 9.80 mm Hg to a postoperative mean of 11.56 ± 2.65 mm Hg (p = 0.001). The mean number of preoperative medications for IOP control was reduced from 3.0 ± 0.7 to 0.4 ± 0.9 (p <0.05) at the end of the follow-up period. No significant improvement in visual acuity was noted from a median pre-operative log MAR of 0.60 ( IQR 0.30 to 1.30 ) to a median log MAR of 0.48 (IQR 0.30 to 1.00 ) at the last follow-up visit.
There were no intraoperative complications. Post operatively, two patients (22%) had choroidal detachments. Although patient 9 subsided with conservative management, patient 4 needed anterior chamber reformation with peripheral anterior synechiae release and tube repositioning in view of post operative shallow chamber.
Discussion
Tube shunt implantation into the posterior chamber sulcus may minimise adverse effects of anterior chamber placement thus, preventing complications associated with surgical vitrectomy during pars-plana insertion. Studies have shown comparable outcomes between sulcus placement and anterior chamber placement of the tube with a higher IOP reduction ratio in the sulcus group.13
To the best of our knowledge, this is the first case series describing this technique of tube shunt placement guided by a 21 G vein needle. The common technique of sulcus implantation was as described by Wiener and colleagues 12 involving Baerveldt (350 mm 2) tube shunt placement. Following creation of a scleral tunnel and using a 23 G needle mounted on a Viscoat syringe, an entry was made and needle was directed towards the ciliary sulcus 1.5 to 2 mm from the limbus. The tube was then inserted through the tunnel and advanced until it emerged behind the iris. Sectoral iridectomy was additionally performed in non dilating pupils. Similarly, Tello et al 9 in their study of 8 eyes described ciliary sulcus insertion of the Barveldt tube shunt through a track created by a 23 G needle 2.5 mm from the limbus. The tube was then placed through the entry of the needle parallel to the posterior iris into the ciliary sulcus following insertion of Sodium hyaluronate (Healon, Advanced Medical Optics, Santa Ana, California, USA) via a temporal paracentesis.
In contrast to these techniques, the main advantage of our ab interno procedure is the accuracy of tube placement in the sulcus in a controlled fashion by withdrawal of the needle. The ease of tube insertion may help shorten the learning curve and enable training surgeons to attain accurate tube placement in the sulcus, thus obviating the need for multiple entry attempts. Possible complications from this technique could be attributed to the large needle size for which, a generous fill of the anterior chamber and sulcus with a highly cohesive viscoelastic is recommended. In our procedure, obtaining the outer diameter specifications of the tube from the manufacturers and procuring a needle of adequate diameter ensured a reasonable fit. The specific 21 G vein needle from the IV set differed from a regular 21 G needle by having a shorter, less sharp bevel thereby limiting damage to the tube.
In the present case series, there were no patients with hyphema or significant post operative inflammation suggesting minimal damage to the angle structures and limited contact of the tube to the iris pigment epithelium with this technique. Significant decrease in IOP and number of AGM was noted in our study similar to others.12 However, owing to the short follow up period, the success rates per se could not be ascertained. The occurrence of choroidal detachments in two patients with the need for reoperation in one could be attributed to the angle closure glaucoma rather than peritubular leakage. Further studies comparing both the methods of tube insertion would be helpful in ascertaining the same.
The limitations to this study is its retrospective nature, small sample size, limited follow-up and lack of corneal endothelial counts prior to the procedure. Using a hollow needle of adequate diameter, this technique is amenable for use with other glaucoma drainage devices as most of them have a silicone tube of identical dimensions to the AADI implant. The effectiveness of this implantation technique could also be better substantiated if compared with the conventional method.
In conclusion, our technique of sulcus placement of the AADI tube is a precise and simple technique of tube insertion. It is a safe alternative to existing procedures limiting multiple blind entries and thereby, preventing damage to adjacent angle structures. Further study is required to validate the long-term safety and efficacy of this technique.
References
- Desai MA, Gedde SJ, Feuer WJ, et al. Practice preferences for glaucoma surgery: a survey of the American glaucoma society in 2008. Ophthalmic Surg Lasers Imaging 2011;42:202–8.
- 2) Pandav SS, Seth NG, Thattaruthody F, Kaur M, Akella M, Vats A, Kaushik S, Raj S. Long-term outcome of low-cost glaucoma drainage device (Aurolab aqueous drainage implant) compared with Ahmed glaucoma valve. Br J Ophthalmol. 2020 Apr;104(4):557-562. 3) Philip R, Chandran P, Aboobacker N, Dhavalikar M, Raman GV. Intermediate-term outcome of Aurolab aqueous drainage implant. Indian J Ophthalmol. 2019 Feb;67(2):233-238.
- 4) Raj S, Jurangal A, Gupta G,Thattaruthody F, Seth N, Panday S. Comparison of Short-term Outcomes of Aurolab Aqueous Drainage Implant with Ahmed Glaucoma Valve in Post–Penetrating Keratoplasty Glaucoma. Ophthalmology Glaucoma. 2019 May-June; 3 (2) : 172 – 177
- Senthil S, Gollakota R, Ali MH , Turaga K, Badakere S, Krishnamurthy R, Garudadri CS. Comparison of the New Low-Cost Nonvalved Glaucoma Drainage Device with Ahmed Glaucoma Valve in Refractory Pediatric Glaucoma in Indian Eyes. Ophthalmology Glaucoma. 2018 Nov-Dec; 3 (1) : 167 – 174
- Puthuran GV, Palmberg PF, Wijesinghe HK, Pallamparthy S, Krishnadas SR, Robin AL. Intermediate-Term Outcomes of an Affordable Aqueous Drainage Implant in Adults with Refractory Glaucoma. Ophthalmology Glaucoma. 2019 Jul; 2 (4) : 258-266.
- Maheshwari D, Dabke S, Rajagopal S, Kadar MA, Ramakrishnan R. Clinical outcome of a nonvalved Aurolab aqueous drainage implant in posterior segment versus anterior chamber. Indian J Ophthalmol. 2019 Aug;67(8):1303-1308.
- Minckler DS, Francis BA, Hodapp EA, et al. Aqueous shunts in glaucoma: a report by the American Academy of Ophthalmology. Ophthalmology 2008; 115:1089 – 1098.
- Tello, C Espana, EM Mora, R Dorairaj, S Liebmann, JM Ritch, R Baerveldt glaucoma implant insertion in the posterior chamber sulcus. Br J Ophthalmol 200791 6: 739–742
- Sidoti PA, Mosny AY, Ritterband DC, et al. Pars plana tube insertion of glaucoma drainage implants and penetrating keratoplasty in patients with coexisting glaucoma and corneal disease. Ophthalmology 2001;108:1050–8.
- Arroyave CP, Scott IU, Fantes FE, et al. Corneal graft survival and intraocular pressure control after penetrating keratoplasty and glaucoma drainage device implantation. Ophthalmology 2001;108:1978–85.
- Weiner, A Cohn, AD Balasubramaniam, M Weiner, AJ Glaucoma tube shunt implantation through the ciliary sulcus in pseudophakic eyes with high risk of corneal decompensation. J Glaucoma 201019 6: 405–411
- Bayer A, Önol M. Clinical outcomes of Ahmed glaucoma valve in anterior chamber versus ciliary sulcus. Eye (Lond). 2017 Apr;31(4):608-614.
Sidoti PA, Mosny AY, Ritterband DC, et al. Pars plana tube insertion of glaucoma
- drainage implants and penetrating keratoplasty in patients with coexisting glaucoma and corneal disease. Ophthalmology 2001;108:1050–8.
- Arroyave CP, Scott IU, Fantes FE, et al. Corneal graft survival and intraocular pressure control after penetrating keratoplasty and glaucoma drainage device implantation. Ophthalmology 2001;108:1978–85.
- Weiner, A Cohn, AD Balasubramaniam, M Weiner, AJ Glaucoma tube shunt implantation through the ciliary sulcus in pseudophakic eyes with high risk of corneal decompensation. J Glaucoma 201019 6: 405–411
- Bayer A, Önol M. Clinical outcomes of Ahmed glaucoma valve in anterior chamber versus ciliary sulcus. Eye (Lond). 2017 Apr;31(4):608-614.
Table 1
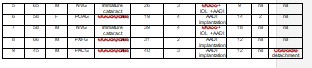
NVG : neovscular glaucoma, PXFG : pseudoexfoliation glaucoma AGM : antiglaucoma medication, Phaco : phacoemulsification, AADI :Aurolab aqueous drainage implant, RD : retinal detachment, ICE : iridocorneal endothelial syndrome, AGM : Antiglaucoma medication
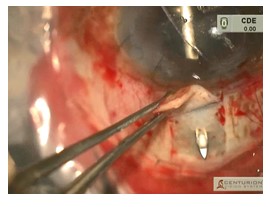
Figure 1 : Needle exiting the scleral bed partially 2.5 mm from the limbus


 FP1050 : A novel ab interno technique of AADI tube sulcus placement : initial outcomes
FP1050 : A novel ab interno technique of AADI tube sulcus placement : initial outcomes
Leave a Comment