Dr. VIVEK DAVE
Dr.Savitri Sharma, Dr.Dimple Lodha, Ms.Roshni Karolia
Abstract
Prospective, comparative, non-randomized, consecutive case series. All cases of endophthalmitis from 1st August 2018 to 31st July 2019 culture positive for bacteria were included and tested in-vitro for biofilm formation by crystal violet and tetrazolium XTT salt method. Cases were classified as biofilm negative (BN) and positive (BP). Anatomic success was defined as globe salvage and functional success was defined as final vision ≥20/200. There were 50 eyes in the BN group and 33 eyes in the BP group. Surgical interventions required were 2.86±1.45 and 6.36±2.89 respectively, (p<0.0001). Final logMAR vision noted was a median of 1.2 and 1.9 respectively (p=0.0005). Functional success was noted in 44% and 21.2% (p=0.03) and anatomic success in 68% and 42.42% respectively (p=0.02). Bacteria with in-vivo biofilm formation in endophthalmitis, show enhanced in-vivo virulence leading to increased need of surgical intervention and a reduction in the final anatomic and functional success.
Full Text
To compare the clinico-microbiological features and outcomes in patients with infectious endophthalmitis caused by biofilm-positive (BP) and biofilm-negative (BN) bacteria.
METHODS
This wasa prospective, interventional, comparative, non-randomized, consecutive case series. Culture-positive bacterial endophthalmitis cases from 1st August 2018 to 31st July 2019 were included. All vitreous samples were tested for biofilm using crystal violet plate and XTT (2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide) methods and classified as BN and BP. Antibiotic susceptibility of all organisms was determined. Anatomic and functional success was defined as intraocular pressure > 5 mm Hg and a final best-corrected vision ≥20/400 respectively at last visit.
RESULTS
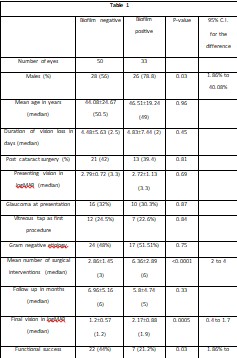
There were 50 eyes in the BN group and 33 eyes in the BP group. BN group eyes required 2.86±1.45 surgical interventions, and BP group eyes needed surgical 6.36±2.89 interventions, p<0.0001, 95% Confidence Interval, CI. 2- 4. Median follow-up was 6 and 5 months, respectively (p=0.33). Final log MAR vision was a median of 1.2 and 1.9 respectively; p=0.0005, 95% C.I. 0.4 to 1.7. Functional success was achieved in 44% and 21.2% (p=0.03, 95% C.I. 1.86% to 40.08%) and anatomic success was achieved in 68% and 42.42% respectively (p=0.02, 95% C.I. 3.85% to 45.47%). The antimicrobial resistance patterns between the two groups were comparable. CONCLUSIONS: Endophthalmitis caused by the biofilm-forming bacteria need a greater number of surgical interventions. The anatomic and functional outcomes are poorer than non-biofilm forming bacterial endophthalmitis. The increased virulence and poorer outcomes can be hypothesized to be due to the physical barrier effect of the biofilm to the antibiotics. KEYWORDS: Biofilm, bacterial endophthalmitis, clinical outcome, endophthalmitis, INTRODUCTION Endophthalmitis is defined as inflammation of the inner coats of the eyeball, primarily involving the vitreous. It is one of the most dreaded eye conditions. Most cases occur either following surgery or trauma. The organisms gain entry either from exogenous sources like trauma orsurgery or from endogenous hematogenous spread from a distant site.[1]Bacterial endophthalmitis is more common than fungal endophthalmitis. Staphylococcus epidermidis is the most common isolate in the USA[ ], Europe[ ], and India.[3] Biofilm is one of the major causes of resistance to various antibiotics in systemic diseases.[4]Structurally, a biofilm is a slimy layer of an extracellular matrix made of polymeric substances produced by microorganisms (Fig 1). This forms an architectural colony providing resistance not only against antibiotics but also against the human immune system.
[5]Role of biofilm has been studied in several ocular conditions where implants are used (such as intraocular lens, scleral buckles, punctal plugs, and lacrimal intubation devices) and not used (such as keratitis, chronic dacryocystitis, and endophthalmitis).[6]Microorganisms produce biofilm by various mechanisms related to biochemical, molecular, and altered host factors.[7]Leid et al demonstrated the development of histologically proven biofilm on the posterior surface of the lens capsule about 72 h after injecting 5000 cfu/ml of S. aureus RN 6390 into the mid-vitreous cavity in a murine model.[8] There are no reports on the impact of biofilm on the clinical management of endophthalmitis and its outcome in existing literature. In the current communication, we present our results of evaluating bacterial biofilm and its role in the management outcome of endophthalmitis. METHODS This was a prospective, comparative, non-randomized, consecutive case series. Patients diagnosed with culture-positive bacterial endophthalmitis from 1st August 2018 to 1st August 2019 were recruited into the study. Data of patients with a minimum follow-up of 3 months were analyzed. The study was approved by the Institutional Review Board (LEC -7-18-118) and adhered to all the tenets of the Declaration of Helsinki on treating human subjects. Written informed consent was obtained from all patients and guardians, when patients were younger than 18 years. The exclusion criteria included all culture-negative cases of infectious endophthalmitis, all cultures positive for fungus, and patients not consenting to the study. All patients underwent a detailed, comprehensive ophthalmic evaluation including uncorrected visual acuity (UCVA), best-corrected visual acuity (BCVA) using Log MAR chart, slit-lamp biomicroscopy, intraocular pressure (IOP) by Goldman applanation tonometry (GAT),
a dilated fundus examination using 78/90/20D lens, and B-Scan ultrasound evaluation when fundus was not visible. Endophthalmitis was diagnosed clinically (and with B-scan ultrasonography, when required) from a combination of symptoms (pain, red eye, lid edema, reduced presenting vision) and signs (corneal edema, exudates in the anterior chamber, hypopyon, vitreous exudates, medium to high reflective dot or membranous opacities in the vitreous cavity and a thickened choroid). Undiluted vitreous was processed in microbiology laboratory for direct microscopy and culture for bacteria and fungi as per the institutional protocol.[9]The bacterial isolates were identified by Vitek 2 compact system (bioMerieux),and antibiotic susceptibility was tested by a combination of E test and Vitek 2 for minimum inhibitory concentration (MIC) of several antibiotics and interpreted as per CLSI (Clinical and Laboratory Standards Institute)guidelines.10The bacterial isolate was then tested for biofilm formation in vitro using crystal violet method11 and XTT (2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide) method.12(Fig 2). Functional success was defined as visual acuity logMAR 1.3 (Snellen >20/400) and anatomical success as IOP >8 mm Hg.[ ]
CLINICAL MANAGEMENT PROTOCOL
As per institute protocol, the surgical management of endophthalmitis consisted of pars plana vitrectomy, microscopy and culture of undiluted vitreous, antimicrobial susceptibility testing of bacterial isolates, intravitreal antibiotics (vancomycin (1 mg/0.01 ml) + ceftazidime (2.25 mg/0.01 ml)) with or without dexamethasone (400 μg/0.01 ml). The medical treatment also included intensive topical antibiotics (ciprofloxacin 0.3% one hourly) and corticosteroid (prednisolone acetate 1% one hourly) and oral ciprofloxacin 750 mg two times per day for 7-10 days. Additional procedures such as repeat intravitreal antibiotics or
repeat pars plana vitreous lavage depended on the response to treatment and were left to the decision of the treating physicians. In principle, intravitreal injections were repeated every 48 hours. Media clarity was recorded in terms of reduction of vitreous echoes on B scan at each subsequent visit and in terms of improvement in the visibility of the retina and retinal vessels. Repeat injections were discontinued once media clarity increased to at least second order retinal vessels visible. In cases with hazy view due to corneal involvement, a vitreous biopsy was taken instead of a vitrectomy procedure. The topical and intravitreal antibiotics used were adjusted as per the culture sensitivity report.
STATISTICAL METHODS
The collected data was arranged on an excel spreadsheet. Statistical analysis was analyzed using the MedCalc Statistical Software version 19.7.2 (MedCalc, Ostend, Belgium). All continuous data were classified as either normative or non-normative in each group. Paired t-test was used to compare normative data, and the Mann-Whitney U test was used to compare non-normative data. Analysis of categorical data was done using the chi-square test. Odd’s ratio was calculated wherever appropriate. Multivariate logistic regression analysis was done to assess the effect of various demographic and clinical factors on the final anatomic and functional outcome. A p-value of <0.05 was assigned as statistically significant.
RESULTS
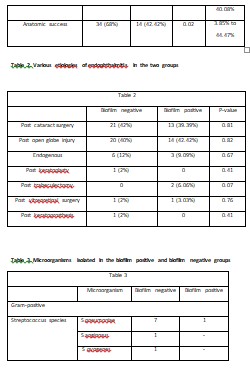
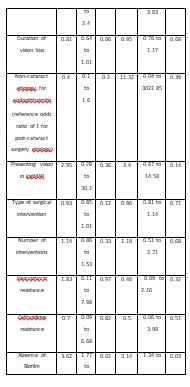
Eighty-three patients satisfied the inclusion criteria in the study period of one year. Fifty patients’ were biofilm positive, and 33 patients were biofilm negative with either CVPor XTT method, or both. Demographic details of the patients in both groups are given in Table 1. There was no statistical difference in the presenting age, duration of vision loss, or presenting vision between the groups. The proportion of males in the two groups were 78.8% and 56% respectively (p= 0.03). The etiology of endophthalmitis was comparable in both groups (Table 2).
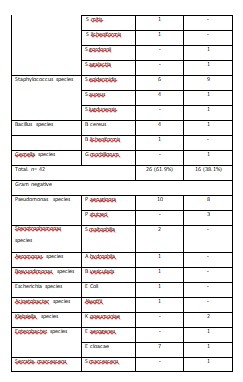
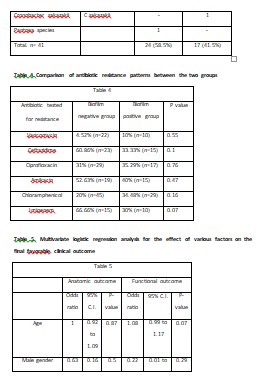
Vitreous biopsy was performed in all patients. It showed a combination of gram-positive and gram-negative organisms (Table 3). In general, biofilm production was similar, 38.1% (n= 16 of 42) in gram-positive bacteria and 41.5% (n= 17 of 41) in gram-negative bacteria; it was seen more often in Staphylococcus spp. (68.7%; n=11 of 16 gram-biofilm producing positive bacteria) and Pseudomonas spp, (64.7%; n=11 of 17 biofilm-producing gram-negative bacteria) (Table 2).The antibiotic resistance pattern is listed in Table 4.
In the negative biofilm group, 34 (68%) eyes met the criteria of anatomical success, and 22 (44%) eyes met the criteria of functional success. Comparatively, in the positive biofilm group, 14 (42.4%) eyes and 7 (21.2%) eyes met the anatomical and functional success criteria, respectively. The biofilm negative and positive group differed in both the anatomical (p=0.02) and functional (p=0.03) success. Further, the eyes with positive biofilm infection needed a statistically greater number of surgical interventions (p <0.0001). Despite an increased number of surgical interventions, final visual acuity remained poor in biofilm-positive endophthalmitis patients. Logistic regression analysis showed no influence of other demographic or clinical factors on the final anatomic or functional outcome. (Table 5).
DISCUSSION
The current study discusses the role of biofilm in the treatment outcome of bacteria endophthalmitis. It was noted that bacteria with in-vivo biofilm formation in endophthalmitis showed enhanced in-vivo virulence leading to an increased need for surgical intervention and reduced final anatomic and functional success. The current study also included cases that were post-trauma. Trauma by itself is a confounding factor that can directly affect final clinical outcomes irrespective of the severity of endophthalmitis. However, as the number of cases post-trauma in both groups was comparable (p=0.82), the effect of this confounding variable was avoided. Griffiths et al. reported biofilm formation on an IOL by S. epidermidis in 1989. 13Adherence of the bacteria to the lens with glycocalyx around it prevents both antibiotics and antibodies from reaching the bacteria.Increased adherence of S. epidermidis to intraocular polypropylene lenses compared to polymethylmethacrylate lenses are known. 14It is proposed that biofilm production on IOLs after cataract surgery occurs due to the microorganism’s ability to adhere to these lenses via exo-polysaccharides produced by them. This provides an extra shell and protects these bacteria from the antibiotics. 15,16Need for multiple interventions and eventually the IOL explanation has also been reported in acute and chronic biofilm-producing Staphylococcal endophthalmitis. 17 However, biofilm formation is not always associated with antibiotic resistance but may affect virulence. Biofilm producing S. epidermidis produces extracellular polymeric substances and inhibits phagocytosis and antibiotics’ action, resulting in inadequate clearance of organisms.18Hence in these situations, multiple interventions may be required to sterilize the vitreous cavity. The current study showed the need for significantly more interventions in biofilm-producing microorganisms causing endophthalmitis as compared to those that were biofilm-negative.
Biofilms have been known to have a virulent role in ophthalmology, especially in cases where some kind of implant has been used for management of the disease. A study conducted by Holand et al, has demonstrated biofilm in 65% of scleral buckles (solid silicone and sponge forms) removed for infection and extrusion by scanning electron microscopy.19 Biofilms on scleral buckles may function as reservoirs for pathogenic bacteria contributing to its extrusion.20 A report by Yokoi N et al, has shown association of biofilm in punctal plugs developing conjunctivitis that required removal of the plug and prolonged antibiotic treatment.21Periorbital implants include orbital plates, porous polyethylene floor implant, orbital sphere implants, anophthalmic socket sphere implants or metal screws. Scanning electron microscopy has demonstrated polymicrobial or mixed species biofilm on these implants. However, most commonly seen organism in orbital implants was S. aureus. Other organisms included M. chelonae, Pantoea agglomerans found in polymicrobial cases, yeasts (Candida spp.,
and Trichosporonspp.), Staphylococcusspp., M. chelonae and Gram-negative bacilli (Achromobacter xylosoxidans and P. aeruginosa) in orbital plates.22Virulence related to biofilm formation has also been proposed in contact lens-related keratitis. One of the commonest organisms implicated in contact lens associated keratitis is Pseudomonas species which is also known to form biofilm. A study conducted by Abidi et al, has shown that all species of Pseudomonas were found to be potential biofilm formers and also concluded that the multi-drug resistant isolates displayed significant biofilm production as compared to susceptible isolates indicating the anti-microbial resistance offered by the biofilm to these organisms.23 Biofilm also forms with Acanthamoeba spp. which is another most common organism involved in contact lens associated keratitis.24
The current study had its strengths and limitations. Among strengths, this was a prospective consecutive case series where the patients were treated by a uniform institutional protocol, but the treating physicians were masked to the results of the biofilm. Among limitations, the biofilm formation was tested in-vitro in cultures and not over IOLs because its explantation was not needed in any of the patients. Thus, our study is an in-vivo extrapolation of the in-vitro observation. Furthermore, the cases in this study also included etiologies other than intraocular surgery, such as open globe injury. This makes the instances heterogeneous, but a logistic regression analysis did not impact the etiological differences on the final clinical outcome.
CONCLUSION
We propose that early identification of biofilm-formingorganisms may help decide a tailored management strategy for each patient. It would also help in proper prognosticating the outcome. These results can serve as a background for further research into anti-biofilm measures impacting clinical outcomes in patients with endophthalmitis.
REFERENCES
1. Han DP, Wisniewski SR, Wilson LA, Barza M, Vine AK, Doft BH, et al. Spectrum and susceptibilities of microbiologic isolates in the Endophthalmitis Vitrectomy Study. Am J Ophthalmol 1996; 122:1‑17.
2. Endophthalmitis Study Group, European Society of Cataract and Refractive Surgeons. Prophylaxis of postoperative endophthalmitis following cataract surgery: Results of the ESCRS multicenter study and identification of risk factors. J Cataract Refract Surg 2007; 33:978‑88.
3. Lalitha P, Sengupta S, Ravindran RD, Sharma S, Joseph J, Ambiya V, et al. A literature review and update on the incidence and microbiology spectrum of postcataract surgery endophthalmitis over past two decades in India. Indian J Ophthalmol 2017; 65:673-77.
4. Das T, Kunimoto DY, Sharma S, Jalali S, Majji AB, Rao TN, et al. and the Endophthalmitis research Group. Relationship between clinical presentation and visual outcome in postoperative and posttraumatic endophthalmitis in South central India. Indian J Ophthalmol 2005; 53: 5-16.
5. Harty DW, Handley PS. Expression of the surface properties of the fibrillar Streptococcus salivarius HB and its adhesion deficient mutants grown in continuous culture under glucose limitation. J Gen Microbiol. 1989;135(10):2611–21.
6. Zegans ME, Becker HI, Budzik J, O’Toole G. The role of bacterial biofilms in ocular infections. DNA Cell Biol. 2002;21(5–6):415–20.
7. Venkatesan N, Perumal G, Doble M. Bacterial resistance in biofilm-associated bacteria. Future Microbiol. 2015;10(11):1743–50.
8. Leid JG, Costerton JW, Shirtliff ME, Gilmore MS, Engelbert M. Immunology of Staphylococcal biofilm infections in the eye: new tools to study biofilm endophthalmitis. DNA Cell Biol. 2002;21(5–6):405–13.
9. Sharma S, Jalali S, Adiraju MV, Gopinathan U, Das T. Sensitivity and predictability of vitreous cytology, biopsy, and membrane filter culture in endophthalmitis. Retina. 1996;16(6):525-9.
10. CLSI, Performance Standards for Antimicrobial Disk Susceptibility Tests. Approved standard, 7th ed. CLSI document M02-A11. Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA. 2012.
11. Corte L, Casagrande Pierantoni D, Tascini C, Roscini L, Cardinali G. Biofilm Specific Activity: A Measure to Quantify Microbial Biofilm. Microorganisms. 2019 Mar 7;7(3):73.
12. Dhale RP, Ghorpade MV, Dharmadhikari CA. Comparison of various methods used to detect biofilm production of Candida species. J Clin Diagn Res. 2014 Nov;8(11):DC18-c20.
13. Griffiths PG, Elliot TS, McTaggart L. Adherence of Staphylococcus epidermidis to intraocular lenses. Br J Ophthalmol. 1989 Jun;73(6):402-6.
14. Elder MJ, Stapleton F, Evans E, Dart JK. Biofilm-related infections in ophthalmology. Eye (Lond). 1995;9 ( Pt 1):102-9.
15. García-Sáenz MC, Arias-Puente A, Fresnadillo-Martinez MJ, Matilla-Rodriguez A. In vitro adhesion of Staphylococcus epidermidis to intraocular lenses. J Cataract Refract Surg. 2000 Nov;26(11):1673-9.
16. Das D, Bhattacharjee H, Gogoi K, Das JK, Misra P, Dhir P, Deka A. Intraocular lens biofilm formation supported by scanning electron microscopy imaging. Indian J Ophthalmol. 2019 Oct;67(10):1708-1709.
17. Pichi F, Nucci P, Baynes K, Carrai P, Srivastava SK, Lowder CY. Acute and chronic Staphylococcus epidermidis post-operative endophthalmitis: The importance of biofilm production. Int Ophthalmol. 2014 Dec;34(6):1267-70.
18. Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: a common cause of persistent infections. Science. 1999 May 21;284(5418):1318-22.
19. Holland SP, Pulido JS, Miller D, Ellis B, Alfonso E, Scott M, Costerton JW. Biofilm and scleral buckle-associated infections. A mechanism for persistence. Ophthalmology. 1991 Jun;98(6):933-8.
20. Zegans ME, Becker HI, Budzik J, O’Toole G. The role of bacterial biofilms in ocular infections. DNA Cell Biol. 2002 May-Jun;21(5-6):415-20.
21. Yokoi N, Okada K, Sugita J, Kinoshita S. Acute conjunctivitis associated with biofilm formation on a punctal plug. Jpn J Ophthalmol. 2000 Sep-Oct;44(5):559-60.
22. Bispo PJ, Haas W, Gilmore MS. Biofilms in infections of the eye. Pathogens. 2015 Mar 23;4(1):111-36.
23. Lalitha P, Sengupta S, Ravindran RD, Sharma S, Joseph J, Ambiya V, Das T. A literature review and update on the incidence and microbiology spectrum of postcataract surgery endophthalmitis over past two decades in India. Indian J Ophthalmol 2017;65:673-7
24. Hashemian H, Mirshahi R, Khodaparast M, Jabbarvand M. Post-cataract surgery endophthalmitis: Brief literature review. J Curr Ophthalmol. 2016;28(3):101-105.
FIGURE LEGENDS
1. Cartoon showing the pathophysiologic cycle of formation of biofilms. The organisms attach on the tissue surface followed by colonization. Colonization is followed by laying down of lipids, nucleic acids, polysaccharides and proteins which form the structure of the biofilm
2. Photograph showing negative and positive controls for biofilm formation by the crystal violet method (Top Panel) and the XXT method (Bottom panel)
Table 1. Comparative presentations and outcomes of biofilm negative and biofilm positive organisms



 FP0413 : Comparative outcomes in endophthalmitis caused by biofilm positive and biofilm negative bacteria
FP0413 : Comparative outcomes in endophthalmitis caused by biofilm positive and biofilm negative bacteria
Leave a Comment